By Kelly Lohr
Imagine slowly losing control of your muscles, first with a few twitches in your arms and legs or a slurred word here or there. Muscle failure will continue until it eventually stops your ability to move, speak, and breathe. This is the life of a patient suffering from amyotrophic lateral sclerosis (ALS) also known as Lou Gehrig’s disease, a progressive neurodegenerative disorder. Currently, there is little treatment for the rapid course of this disease, but James Weimann, PhD, of Stanford Medical School provides a new hope.
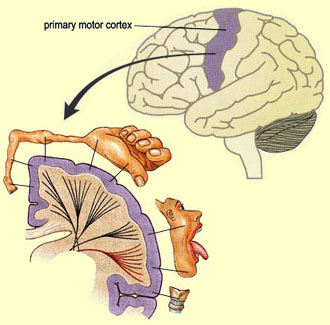
Weimann is part of a team of neuroscientists using transplanted neurons grown from embryonic stem cells to replace damaged cells in young animals. This finding is the first of its kind in that the stem cells can be directed to take on the jobs of specific brain cells while also making the correct connections with other cells. Weimann’s cells transmit information from the cortex, the neural tissue that is outermost part the mammalian brain, specifically areas needed for motor function.
Up until this point, the issue of stem cell transplantation in the brain was making the proper neuronal connections. As an adult organism, creating the accurate connections in the nervous is extremely complex. During development, superfluous neural connections deteriorate with lack of use. Only the pathways with the most activity remain in adulthood. The chemical or physical signals that once lead the way in development are no longer present. Without such cues, it is difficult for neurons to reach their target areas. For example, the stem cells created in Weimann’s lab must make connections with motor cortex in order to be an effective treatment for disorders like ALS or a traumatic brain injury. Incorrect connections could result in further erratic brain function.

While Weimann’s work holds a lot of potential for further progress and treatments, the studies have involved transplantation in young animal models. Since the majority of neurodegeneration takes place in older adults, the next step will be to explore stem cell transplantation in adult animals. Weimann and his team are hopeful that these newest findings will soon be used in treatment of neurons that are lost or damaged due to spinal cord injuries or diseases like ALS.



