Cholesterol and matrisome networks in Alzheimer’s disease
Alzheimer’s Afternoons Seminar Series (April 16)
Dr. Julia TCW is an Instructor at the Icahn School of Medicine at Mount Sinai in New York (USA). She uses human induced pluripotent stem cells (iPSC), transgenic mouse models, and human tissue samples to explore the effects of the apoe4 risk gene on gene expression and metabolism. Her talk touches on several different projects, including some related to the following open-access articles:
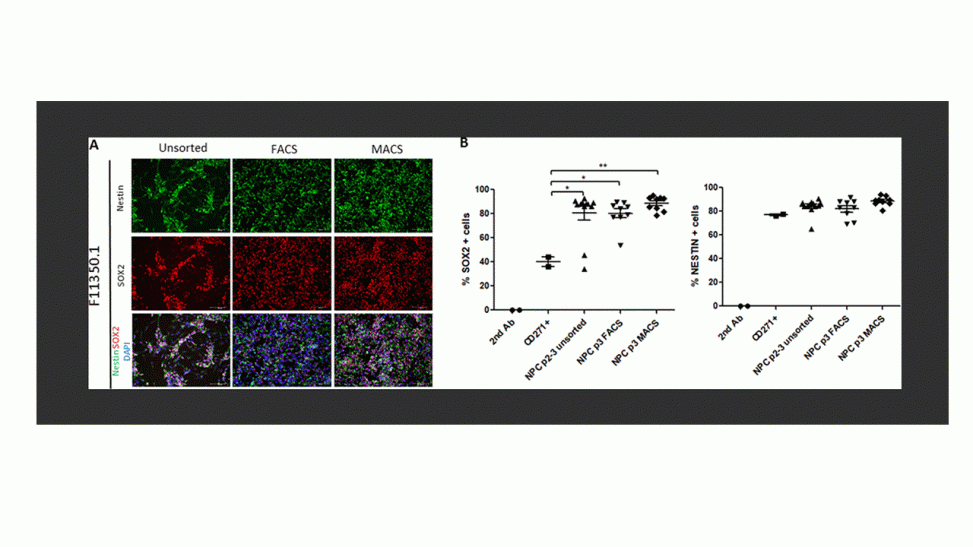
Reduced variability of neural progenitor cells and improved purity of neuronal cultures using magnetic activated cell sorting. Bowles KR, Tcw J, Qian L, Jadow BM, Goate AM. PLoS One. 2019 Mar 27;14(3):e0213374. doi: 10.1371/journal.pone.0213374. eCollection 2019. Erratum in: PLoS One. 2019 Apr 25;14(4):e0216312.
Human iPSC application in Alzheimer’s disease and Tau-related neurodegenerative diseases. Tcw J. Neurosci Lett. 2019 Apr 23;699:31-40. doi: 10.1016/j.neulet.2019.01.043. Epub 2019 Jan 24. Review.
Dr. TCW began with a review of microglia and astrocytes. She pointed out that both microglia and astrocytes react to nearby amyloid beta (Aβ) in the brain by changing the expression of their genes. She focused on two important processes, astrocyte clearance of Aβ and the export of cholesterol from these cells, which depend in some way or another on the apoe protein.
She reminded listeners that the apoe4 variant is the strongest genetic risk factor for late onset Alzheimer’s disease (LOAD). She cited a very recent study on genetic risk:
Andrews, S. J.; Fulton-Howard, B.; Goate, A. Interpretation of Risk Loci from Genome-Wide Association Studies of Alzheimer’s Disease. The Lancet Neurology 2020, 19 (4), 326–335. https://doi.org/10.1016/S1474-4422(19)30435-1.
She mentioned that this study concluded that apoe4/apoe4 individuals have a 15-fold higher lifetime risk of LOAD. This is consistent with previous estimates.
After covering some of the basics of apoe4 protein structure and function, she emphasized that in the brain apoe is produced mainly by microglia and astrocyctes.
She studied the impact of 4/4 vs 3/3 genotypes in three types of models: human iPSC grown in culture, transgenic mice carrying human genes, and postmortem brain tissues from human subjects.
NOTE: it is relatively rare to find studies which focus on apoe4/apoe4 homozygotes. Most studies struggle to find enough human 4/4 carries for studies and, as a result, group 4/4s and the more common 3/4s together as “apoe4 carriers”, a shortcut bound to muddy the data. This clear distinction, in three different models, is a real strength of these studies.
When making human iPSC lines, tissue samples were obtained from human 3/3 or 4/4 subjects. Cells were converted from fibroblasts, to stem cells, to neural progenitor cells, and then to neurons and astrocytes. Microglia can be produced along the way as well. The process, which takes les than 30 days, is described in the following open-access article:
An Efficient Platform for Astrocyte Differentiation from Human Induced Pluripotent Stem Cells. Tcw J, Wang M, Pimenova AA, Bowles KR, Hartley BJ, Lacin E, Machlovi SI, Abdelaal R, Karch CM, Phatnani H, Slesinger PA, Zhang B, Goate AM, Brennand KJ. Stem Cell Reports. 2017 Aug 8;9(2):600-614. Doi: 10.1016/j.stemcr.2017.06.018. Epub 2017 Jul 27.
The group verified that the apoe4 genotype was the only genetic contributor to AD pathology in these cells. The presence of other risk genes was ruled out. Here they compared 6 vs 7 lines of cells derived from brain tissue of age-matched, caucasian subjects with either apoe3/3 or apoe4/4 genotypes. The age of the donors was not a factor, she indicated, since iPS cells have the typical aging factors removed during the differentiation process. Finally, Dr. TCW noted that there are different types of astrocyctes and they controlled for this in their studies.
They found that:
- In apoe 4/4 microglia pathways associated with cholesterol, lipid, and steroid biosynthesis were increased while pathways assocaited with lysosomal recycling and cholesterol efflux were decreased.
- In apoe 4/4 astrocytes, cholesterol biosynthesis pathway genes were also increased.
- In these apoe4/4 cells several large pathway networks seemed to be altered: SREBF2 and SCAP up, POR down.
- HDL-mediated lipid transport network was inhibited in apoe4/4 microglia (mainly driven by decreased FXR/RXR activity).
As a result, we would expect cholesterol to be accumulating, perhaps to excess, in these cells which are likely to be producing it faster than they export it.
Dr. TCW displayed a diagram of CHO transport throughout cells, it illustrated the familiar process:
Cholesterol is imported by cells as LDL apolipoprotein particles bind to LDL surface receptors and are engulfed into early endosomal “bubbles” in the cytoplasm. Later, endosomal “sorting” occurs in late endosomes, before or after they combine with lysosomes. This process separates cholesterol molecules from the LDL particles, which in turn have been separated from the LDL receptors. The receptors and LDL particles can be recycled back to the cell membrane and released while CHO reaches the endoplasmic reticulum.
Excess cholesterol, beyond what can be used by the cell, forms lipid droplets (perhaps similar to those first observed by Alois Alzheimer’s in postmortem brain tissues). This cholesterol can be exported from the cell when ABCA1 proteins, located on cell membranes, load cholesterol into lipoprotein particles, which include apoe proteins on their surfaces.
Defects in various steps can cause problems. Altered binding of LDL particles to LDL receptors can impact cholesterol import. Altered sorting or disrupted release of complexes within late endosomes can create virtual traffic jams. Defects in ABCA1 and apoe proteins can inhibit cholesterol export.
Dr. TCW then introduced the concept of “matrisome pathway dysregulation” which they have observed in hiPSC mixed cultures.
As a reminder, the matrisome is the collection of proteins that are secreted from cells to form the extracellular matrix. This includes many glycoproteins and collagens. The matrisome is the subset of the larger secretome, the set of all proteins secreted into the extracellular space.
In a series of experiments the researchers examined how apoe 4/4 cells differed with regards to the matrisome and other gene networks. They used three different approaches:
- They examined five different cell types, including microglia and astrocytes, from brains of AD patients (obtained from Mt. Sinai) having apoe 3/3 and apoe 4/4 genotypes.
- They analyzed the matrisome in brain cells from mice – including apoe3/3, apoe4/4, and apoe knockout (KO) mice.
- They also examined this in human cell lines with altered using CRIPSR cas9 technology.
They found that apoe4 causes lipid and cholesterol dysregulation. For instance:
- Cholesterol production was often increased:
- Apoe 4/4 microglia upregulate the synthesis of cholesterol.
- Apoe4/4 astrocytes exhibit a 20% increase in free cholesterol.
- Lipoprotein cholesterol was often higher in apoe 4/4 cultures.
- The activity of HMG CoA reductase, the rate limiting step of cholesterol biosynthesis, was increased in apoe4/4 cells.
- Imaging revealed higher levels of cholesterol in apoe4/4 astrocytes.
- Cholesterol export was often inhibited
- Apoe4/4 cells often displayed decreased cholesterol efflux, levels of apoe protein, and level of ABCA1 protein.
- Apoe4/4 astrocytes displayed higher matrisome network activity, and increases in interferon and cytokines (IL-6 and IL-8) in astrocytes and/or microglia.
These numerous experiments are complicated and generated large amounts of data. For those with interest, it’s worth reading the original papers to learn more.
Taken together, the data seem to indicate that apoe4 causes changes in cholesterol metabolism, inflammation, and the construction of the extracellular matrix, to different degrees in a variety of brain cell types. Dr. TCW concluded by describing three projects which build on these observations.